
Since the FDA-approval of Spravato on March 5, 2019, we have received numerous questions about the drug from those suffering from depression who have not had success with conventional treatments.
Below is a list of the most commonly asked questions about Spravato from our readers, answered by our pharmacist. If you don't find what you are looking for in this article, be sure to ask us directly and we will update this FAQ with your answer!
What Is Spravato (Esketamine)?

Spravato is an intranasal spray containing the active ingredient esketamine hydrochloride.
It is FDA-approved for the treatment of 'treatment-resistant depression' (TRD), or depression that has not responded to conventional antidepressant therapy (such as SSRI drugs). There is no single definition for TRD, but as it concerns Spravato, participants in clinical trials had previously not had benefit from at least two antidepressants.
Who Is Spravato For?

Spravato is indicated by the FDA for the treatment of treatment-resistant depression (TRD) in adults in conjunction with an oral antidepressant.
In other words, in addition to using Spravato, you should also be taking an oral antidepressant, preferably one that you have not tried before. Oral antidepressants that can be used in conjunction with Spravato include:
- SSRIs (e.g. Zoloft, Prozac, Paxil)
- SNRIs (e.g. Cymbalta, Effexor)
- Wellbutrin
Spravato is not indicated for use in children.
How Is Spravato (Esketamine) Different Than Ketamine?
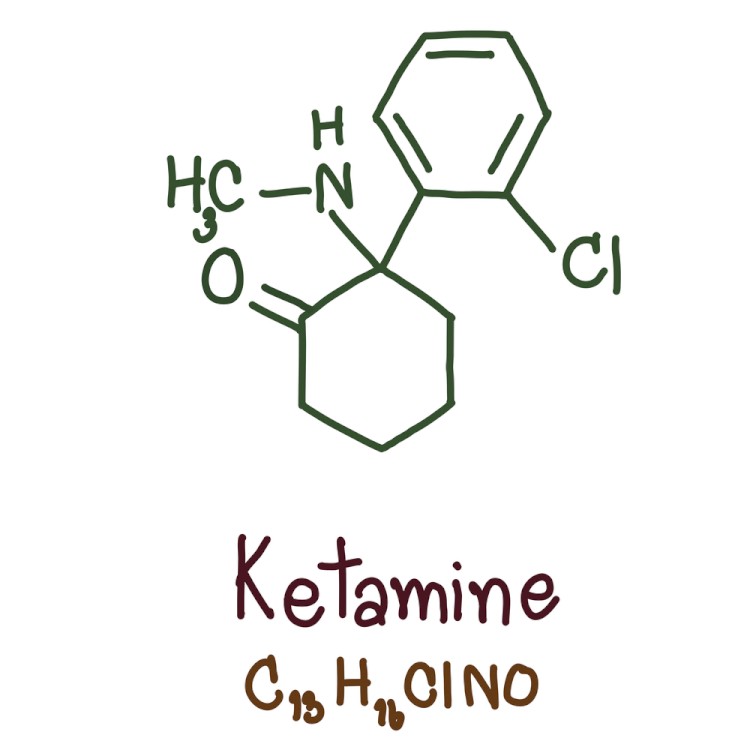
Ketamine is a well-known drug that was originally approved by the FDA in 1970 (as the brand name Ketalar). It is used most commonly as a sedative-hypnotic anesthesia agent for diagnostic and surgical procedures.
Ketamine is a racemic mixture (50-50 mix), and contains two isomers, or molecules that are mirror images of one another:
- Esketamine (also known as 'S-ketamine')
- Arketamine (also known as 'R-ketamine')
Esketamine is simply 'one-half' of ketamine (the left-handed isomer).
So, if you have ever been administered ketamine, you were actually taking 50% esketamine and 50% arketamine.

Drugs that are simply enantiomers of a particular compound are quite common. One example is Nexium (esomeprazole), which is the 'left-handed' isomer of Prilosec (omeprazole).
Enantiomers of drugs are often produced as unique products due to potentially favorable characteristics when compared to their parent compound.
In the case of Nexium (esomeprazole), it reaches far higher concentrations in the body when compared to Prilosec (omeprazole) since it is eliminated more slowly from the body.
Going back to esketamine, most studies show that it is more potent than arketamine (one study reports that it is about twice as potent), but is eliminated more quickly.
How Does Spravato Work?

Spravato (esketamine) works differently than conventional antidepressants (which alter concentrations of various neurotransmitters including dopamine, serotonin, and norepinephrine).
Esketamine is classified as a non-selective, non-competitive antagonist of the N-methyl-D-aspartate (NMDA) receptor. In other words, it is an NMDA-receptor antagonist. How this action translates to an antidepressant effect isn't well understood. The prescribing information for Spravato says as much:
"Esketamine, the S-enantiomer of racemic ketamine, is a non-selective, non-competitive antagonist of the N-methyl-D-aspartate (NMDA) receptor, an ionotropic glutamate receptor. The mechanism by which esketamine exerts its antidepressant effect is unknown."
Although we don't exactly know how ketamine works as an antidepressant, there have been numerous studies exploring its effects.
One study notes specifically that while the overall functions of NMDA receptors are complicated, they likely play an important role in learning, memory, pain perception and neuronal excitability. Drugs that affect NMDA receptors are thought to trigger persistent biochemical and structural changes in the brain.
How Is Spravato Available?
Spravato is available only as a nasal spray, and each spray device delivers two sprays containing a total of 28 mg of esketamine.
Depending on your prescribed dose, you may be using more than one bottle of nasal spray per treatment.
Can I Get Spravato At My Pharmacy?

Spravato is not available at your local pharmacy. It must be used at your doctor's office, under the direct supervision of a healthcare provider.
In fact, due to the potential risk of illicit use (it is classified as a schedule III controlled substance) and since it can cause side effects that must be monitored for, Spravato is only available as part of a REMS (Risk Evaluation and Mitigation Strategies) program.
REMS programs are designed to educate both practitioners and patients on the risk of severe side effects as well as provide conditions for dispensing. For Spravato, the REMS program is in place due to the risks of blood pressure increases, sedation, dissociation, and abuse/ misuse. The requirements are:
- Healthcare settings must be certified to dispense and prescribe Spravato.
- Spravato can only be dispensed in healthcare settings.
- Patients must be enrolled in the REMS program and educated on the risks of the drug.
- Spravato can only be used under the direct observation of a healthcare provider.
- After administering Spravato, individuals must be monitored by a healthcare provider for at least 2 hours after administration.
What Is The Dosing For Spravato?
Spravato is only indicated to be used along with an oral antidepressant.
The recommended dosing schedule is done in two phases:
- Induction Phase
- Maintenance Phase
Induction Phase
- Week One to Four: Administer twice a week. The first dose is recommended to be 56 mg while subsequent doses can be 56 mg to 84 mg, based on individual response.
Maintenance Phase
- Week Five to Eight: Administer once weekly (56 mg or 84 mg based on individual response).
- Week 9 and After: Administer 56 mg to 84 mg every one to two weeks (based on individual response).
This is summarized in the table below.

How Fast Does It Work?
One of the potential benefits of Spravato is how quickly it can work.
Unlike traditional antidepressants, like SSRIs and SNRIs, which can take up to 4 to 6 weeks to notice initial effects, Spravato can start working in as little as a few hours.
Studies evaluating the effects of intravenous ketamine have noted that antidepressant and anti-suicidal response rates to the drug were seen as soon as 4.5 hours after a single dose.
The FDA news release announcing the approval of Spravato states that the drug showed a statistically significant improvement in depressive symptoms in just two days:
"...Spravato nasal spray demonstrated statistically significant effect compared to placebo on the severity of depression, and some effect was seen within two days."
How Long Does Spravato Work?

Although longer-term studies are needed, it appears Spravato works as long as you continue on the medication.
Following the recommended dosing schedule, one study that lasted for four weeks noted an improvement in depressive symptoms at the end of the trial compared to those who received a placebo and an oral antidepressant.
A press release from Janssen, the manufacturer of Spravato, states:
"In a long-term study, patients in stable remission taking SPRAVATO™ who continued treatment with the medicine were 51 percent less likely to relapse versus those who maintained a regimen of a placebo and an oral antidepressant."
Is Spravato Safe?
Spravato has a number of reported side effects, and serious ones are possible. Due to this, after Spravato is administered at the doctor's office, individuals must be monitored for at least 2 hours at each treatment session.
The most commonly reported side effects of Spravato are:
- Dissociation
- Dizziness
- Nausea
- Sedation
- Vertigo
- Reduced sense of touch
- Anxiety
- Lethargy
- Increased blood pressure
- Vomiting
- Feeling drunk
Some of the more concerning side effects of Spravato are increases in blood pressure, sedation, and disassociation. It is the potential severity of these three side effects that necessitate individuals using the drug to stay at the doctor's office for monitoring for at least 2 hours.
Sedation
The majority of individuals using Spravato develop some sort of sedation after taking the drug. The prescribing information states an incidence rate between 49% to 61%.
Blood Pressure
Blood pressure spikes are concerning with Spravato. Clinical trials reported that 8% to 17% of individuals experienced an increase of more than 40 mmHg in systolic blood pressure and/or 25 mmHg in diastolic blood pressure in the first one and one-half hours after administration.
Therefore, blood pressure must be checked prior to dosing with Spravato and if your blood pressure is greater than 140/90 mmHg, caution is recommended before administering. You must consider the risks of short term increases in blood pressure versus the benefit of the drug.
Studies show that blood pressure increases peak about 40 minutes after taking Spravato and can last up to four hours.
The prescribing information for Spravato states that if your blood pressure is decreasing and clinically stable after two hours, it should be safe to be discharged.
Disassociation
Ketamine is well known to cause dissociative feelings and Spravato (esketamine) is no different. The incidence rate of this side effect is between 61% and 75%.
Dissociative changes include:
- Distortion of time, space and illusion
- Derealization
- Depersonalization
Additional Information
It needs to be noted that Spravato has limited safety data and the effects of long-term use aren't well known. One of the (dissenting) members of the FDA panel that approved the drug detailed their concerns in a blog post.
How Much Does Spravato Cost?
If you are paying out of pocket for Spravato, the cost is reported to be between $590 to $885 per treatment. The price will vary depending on the dosage taken as you may need to use multiple nasal spray bottles per treatment.
If you are using insurance, your copay will vary by your plan.
Should I Know Anything Before Going to the MD for Spravato?
The prescribing information recommends to:
- Avoid food at least 2 hours before administration.
- Avoid drinking liquids at least 30 minutes prior to administration.
Avoiding food and drink for the periods noted above can help to reduce nausea and vomiting after administering Spravato.
Does It Interact With Other Drugs
Spravato has several drug interactions to be aware of, including:
- CNS depressants (e.g., benzodiazepines, opioids, alcohol) as these may increase sedation
- Stimulants (e.g. amphetamines) and other drugs that may increase blood pressure
- Monoamine Oxidase Inhibitors (MAOIs)
- Spravato may decrease concentrations of drugs metabolized by CYP2B6 and CYP3A4, but further studies are needed to know the potential severity.
Spravato hasn't been thoroughly evaluated for drug interactions so be sure to ask your pharmacist or doctor before combining it with other prescription or OTC drugs.
Is Spravato A Controlled Substance?
Yes, Spravato is a Schedule III controlled substance under the Controlled Substances Act.
References
- Spravato Prescribing Information
- Attenuation of Antidepressant Effects of Ketamine by Opioid Receptor Antagonism. American Journal of Psychiatry
- The clinical use of S-(+)-ketamine--a determination of its place. PubMed
- NMDA receptor antagonists for depression: Critical considerations. PubMed
- Treatment-Resistant Major Depression: Rationale for NMDA Receptors as Targets and Nitrous Oxide as Therapy. PubMed
- FDA approves new nasal spray medication for treatment-resistant depression; available only at a certified doctor’s office or clinic. FDA
- Pharmacokinetic studies with esomeprazole, the (S)-isomer of omeprazole. PubMed




